
Anne Hollis-Coates is out to help young sufferers of Spinal Muscular Atrophy (SMA).
Subscribe now for unlimited access.
$0/
(min cost $0)
or signup to continue reading
Her mission came about after a close friend’s son was diagnosed with the disease shortly after his second birthday.
The young boy had started crawling, but soon began to deteriorate. He has never been able to walk.
Every year in Australia roughly 30 babies are born with SMA, triggered by a mutation on their fifth chromosome that interrupts the production of a particular protein critical for muscle development.
The nerves of the spinal cord deteriorate, atrophy and eventually die. SMA not only wastes away the muscles in arms and legs, but internal muscles used for coughing, swallowing and breathing. There is no cure.
There is four types of SMA, with Type 1 being the most severe. The disease is the biggest killer of babies under two years of age.
Ms Hollis-Coates recently addressed federal parliament, with the hopes of getting Nuscinursen (Spinraza) on the Pharmaceutical Benefit Scheme list.
This antisense drug interferes with the mutation's effect on motor neurones, boosting the level of SMN protein in the central nervous system.
“At the moment this drug is thousands of dollars but we need to make it available for more people,” the Orient Point woman said.
“This medication slows the disease down and delays deterioration which buys time for more research.”
SMA1 babies can access the treatment via drug manufacturer Biogen at no cost, but children with other SMA subtypes can't access the unapproved drug, which costs roughly $US125,000 per dose, and between $US650,000 (AU$850,000) and $750,000 ($A976,000) for one course of treatment – five to six doses over a year.
Spearheaded by support and awareness group SMA Australia, families, SMA clinicians and researchers have three demands:
- Subsidise preconception carrier screening so would-be parents can find out if they are at risk of having a baby with SMA;
- Add SMA to the newborn screening program so the condition can be detected at birth;
- List Nusinersen on the PBS so all families can access the treatment.
Nusinersen has now been approved in Japan and Canada as well as the European Union and the US. The Therapeutics Goods Administration is currently considering an application from Biogen to approve or reject registration for the drug. SMA advocates hope a PBS listing will follow, with political backing. The earliest the drug could be listed is April 2018.
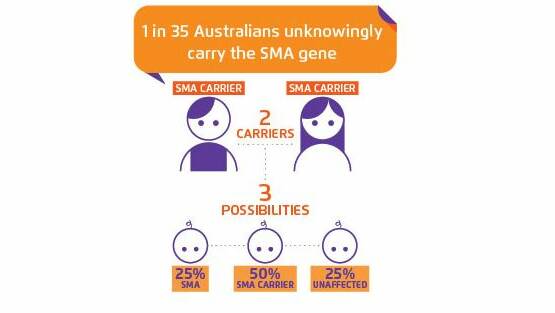
Roughly one in 40 people carry the SMA mutation. If both would-be parents are carriers, they have a one in four chance of having a baby with SMA.
The American College of Obstetricians and Gynaecologists recently recommended obstetricians, gynaecologists and other related healthcare providers offer pre-pregnancy carrier screening for genetic diseases to all patients.
Parents may opt to undergo IVF to select an unaffected embryo.
Carrier testing for SMA and other rare genetic conditions costs between $350 and $1000. The tests are not routinely offered and few parents are aware of the conditions, let alone the tests.
“Nothing prepares parents for when their baby is born with SMA,” Ms Hollis-Coates said.
“We need more education for both the public and the medical fraternity around what tests are available.”
Timely diagnosis is also critical, not only to allow families to process their new reality, but to achieve better outcomes with treatment.
Nusinersen is most beneficial when started early, before symptoms emerge and SMA advocates are pushing for the condition to be added to the newborn screening program, the heel-prick test all newborn babies undergo.
It has been more than 15 years since a condition was added to the newborn bloodspot screening program. But a national framework for newborn screening is currently being considered by the federal government's Standing Committee on Screening (SCoS).
The new framework is expected to be approved by the end of 2017, a spokeswoman for the Department of Health says. SMA Advocates are hopeful SMA will be added to the heel-prick test once the framework is in place.
SMA Australia chief executive Julie Cini has devoted her life to supporting families of children with SMA since her daughters, Montana and Zarlee, died of SMA1 aged 10 and 12 months respectively over a decade ago.
"My biggest concern is to make sure the government knows what SMA is, because they really can do what needs to be done to make sure other families don't go through what I did," she said.
"We'll keep fighting until there is change in Australia for SMA."

